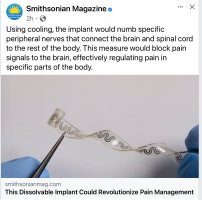
The team created what Rogers describes as a “rubber band” with tiny channels slightly larger than a human hair embedded in it. One side of the 5-millimeter-wide device ends in a cuff-like structure that encircles a nerve. The other comes out of the skin and attaches to a pumping device–similar to how an IV works. Cooling fluid, which boils at a low temperature, is pumped into one of the band’s thin channels to the nerve. It meets up with dry nitrogen that flows through a separate channel, and immediately evaporates, creating a cooling effect. The gas flows back out through a different channel, then recondenses and passes back through the device again, forming a closed loop system. The device is described in a new study published today in
Science.
Inside the device is a tiny temperature sensor, so a user can monitor and control the temperature of the nerve by adjusting the flow rate of the coolant. Getting a nerve too cool can result in tissue damage. The temperature sensor is made up of four layers; a layer of magnesium is encapsulated by two layers of silicon dioxide, an insulating material, and a layer below that serves as an adhesive.
“Current can flow through that magnesium layer—it's a metal—and the resistance of that metal changes as a function of temperature,” Rogers explains.
The implant has been tested in rats, but researchers envision the device could be used in humans in the future. Northwestern University
The device is also fully dissolvable in the body, which eliminates the risks involved in surgical removal. The time it takes to dissolve—usually days or weeks—depends on the material used and its thickness.
After some success on rats, researchers are hopeful this device could provide humans a more targeted and less addictive alternative to opioids

www.smithsonianmag.com

/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer_public/7c/dd/7cdd7672-4f09-46d2-b8ef-d74b5ac7e397/arm_implant_illustration.jpg)
/https://tf-cmsv2-smithsonianmag-media.s3.amazonaws.com/filer_public/12/88/1288d739-90a8-4a00-b378-86e965d01144/img_4419.jpg)

